Definitions [5]
When one or more hydrogen atoms of aliphatic or aromatic hydrocarbons are replaced by a corresponding number of halogen atoms (fluorine, chlorine, bromine or iodine), the resulting compounds are called halogen derivatives of alkanes or arenes.
Define the following:
Chirality
The relationship between a chiral molecule and its mirror image is similar to the relationship between left and right hands. Therefore it is called handedness or chirality
Define the following:
Plane polarized light
A light having oscillations only in one plane perpendicular to direction of propagation of light is known as plane polarized light.
Dehydrohalogenation of alkyl halide is a β-elimination reaction in which the halogen atom is lost from the α-carbon atom and the hydrogen from the β-carbon (according to the Saytzeff rule).
Define organometallic compound.
Active metals like sodium, magnesium cadmium readily combine with alkyl chlorides, bromides and iodides to form compounds containing carbon-metal bonds. These are known as organometallic compounds.
Key Points
1. On the Basis of Hydrocarbon Skeleton
| Type | General Formula | Description |
|---|---|---|
| Haloalkanes | R–X | Halogen attached to an alkane |
| Haloalkenes | R–CH=CHX | Halogen attached to alkene |
| Haloalkynes | R–C≡C–X | Halogen attached to alkyne |
| Haloarenes | Ar–X | Halogen attached to aromatic ring |
2. On the Basis of the Number of Halogen Atoms
| Type | Description |
|---|---|
| Monohalogen | One halogen atom |
| Dihalogen | Two halogen atoms |
| Trihalogen | Three halogen atoms |
| Polyhalogen | More than three halogens |
3. On the Basis of the Nature of the C–X Bond
(A) Compounds containing sp³ C–X bond
| Type | Description | Example |
|---|---|---|
| Alkyl halide | Halogen attached to the sp³ carbon of the alkyl group | R–CH₂–X |
| Allylic halide | Halogen on carbon next to a double bond | CH₂=CH–CH₂–X |
| Benzylic halide | Halogen on carbon next to the benzene ring | C₆H₅–CH₂–X |
(B) Compounds containing a sp² C–X bond
| Type | Description | Example |
|---|---|---|
| Vinylic halide | Halogen directly on the double bond carbon | CH₂=CH–X |
| Aryl halide | Halogen directly on the aromatic ring | Ar–X |
4. Classification of Alkyl Halides
| Type | Structure | Description |
|---|---|---|
| Primary (1°) | R–CH₂–X | Halogen attached to 1 carbon |
| Secondary (2°) | R–CHX–R | Halogen attached to 2 carbons |
| Tertiary (3°) | R₃C–X | Halogen attached to 3 carbons |
Basic rules
- Alkyl halides → haloalkanes (IUPAC)
- Aryl halides → haloarenes
- Longest chain selection
- Numbering for the lowest position
- Substituents named alphabetically
| Formula | Common name | IUPAC name |
|---|---|---|
| CH₃CH₂Br | Ethyl bromide | Bromoethane |
| CH₃CH(Cl)CH₃ | Isopropyl chloride | 2-Chloropropane |
| (CH₃)₂CH–CH₂Br | Isobutyl bromide | 1-Bromo-2-methylpropane |
| (CH₃)₃CBr | Tert-butyl bromide | 2-Bromo-2-methylpropane |
| (CH₃)₃CCH₂Cl | Neopentyl chloride | 1-Chloro-2,2-dimethylpropane |
| CH≡C–Cl | Chloroacetylene | Chloroethyne |
 |
Benzyl iodide | Iodophenylmethane |
 |
p-Iodotoluene | 1-Iodo-4-methylbenzene or 4-Iodotoluene |
- From alkanes
Free radical halogenation:
\[CH_{3}CH_{2}CH_{2}CH_{3}\xrightarrow{Cl_{2}UV-light}CH_{3}CH_{2}CH_{2}CH_{2}Cl+CH_{3}CH_{2}CH(Cl)CH_{3}\] - From alkenes
Addition of hydrogen halide:
C=C + HX → R–X - From alcohols
\[3ROH+PCl_{3}\longrightarrow3R-Cl+H_{3}PO_{3}\]
\[ROH+PCl_{5}\longrightarrow R\longrightarrow Cl+POCl_{3}+HCl\]
\[ROH+HCl\xrightarrow{ZnCl_{2}}R-Cl+H_{2}O\]
\(ROH\xrightarrow{RedP/Cl_{2}}R-Cl\)
\[R—OH+SOCl_2\xrightarrow{\Delta}R—Cl+SO_2\uparrow+HCl\] - Halogen exchange reactions
Finkelstein reaction:
\[R—X+NaI\xrightarrow{Acetone}R—I+NaX\]
Swart's reaction:
\[RBr+AgF\longrightarrow R—F+AgBr\] - From aromatic amines
Sandmeyer reaction:
\[\mathrm{C_{6}H_{5}NH_{2}}\xrightarrow{\mathrm{NaNO_{2}+HX}}\mathrm{C_{6}H_{5}N_{2}^{+}\bar{X}}\xrightarrow{\mathrm{Cu_{2}X_{2}}}\mathrm{C_{6}H_{5}X+N_{2}}\]
- Alkyl halides are colourless when pure but develop colour on exposure to light; they have a sweet smell
- Polarity: Moderately polar
- Solubility: Insoluble in water, soluble in organic solvents
Bond Length
- Increases in order: R–F < R–Cl < R–Br < R–I
Bond Strength
- Decreases in order: R–F > R–Cl > R–Br > R–I
Boiling Point
- Order: R–I > R–Br > R–Cl > R–F
- Increases with an increase in molecular mass
- Decreases with branching
Dipole Moment
-
Decreases as the electronegativity of halogen decreases
Isomeric Dihalobenzenes
- Boiling point: nearly the same
- Melting point: para > ortho/meta
- Optical activity: Ability of a compound to rotate plane-polarised light
- Dextrorotatory (+): rotates light to the right
- Laevorotatory (–): rotates light to the left
- (+) and (–) forms are called optical isomers, and the phenomenon is optical isomerism
- Number of optical isomers = 2ⁿ
(n = number of asymmetric carbon atoms) - Asymmetric carbon/stereocentre: Carbon attached to four different groups
- Chirality: Property of a molecule whose mirror image is non-superimposable
- Enantiomers: Stereoisomers which are non-superimposable mirror images of each other
- Retention: Preservation of spatial arrangement around the asymmetric carbon during reaction
- Inversion of configuration: In the SN2 reaction, backside attack gives a product with the opposite configuration. This complete inversion is called Walden inversion
- Racemic mixture: Equimolar mixture of dextro and laevo isomers. It is optically inactive due to external compensation. This phenomenon is called racemisation.
Representation of configuration:
Fischer projection:
- Horizontal bonds → above plane
- Vertical bonds → below plane
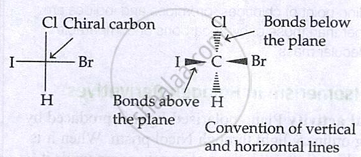
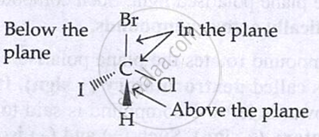
Wedge formula:
- Solid wedge → above plane
- Dashed wedge → below plane
- Straight line → in plane
Alkyl halides mainly undergo substitution and elimination reactions.
1. Nucleophilic Substitution Reactions
SN1 (Unimolecular Nucleophilic Substitution):
- Occurs in two steps:
- Formation of carbocation
- Attack by nucleophile
- First-order reaction (depends only on alkyl halide)
- Reactivity order:
Benzylic > 3° > 2° > 1° > methyl - Leads to racemisation
SN2 (Bimolecular Nucleophilic Substitution):
- Occurs in one step (concerted mechanism)
- Second-order reaction (depends on alkyl halide and nucleophile)
- Reactivity order:
Methyl > 1° > 2° > 3° - Strong nucleophiles increase the rate
- Leads to inversion of configuration
2. Reactions of Alkyl Halides
- With aqueous KOH → Alcohol
- With alcoholic KOH → Alkene (elimination)
- With NaOR → Ether
- With NH₃ → Amine
- With KCN → Nitrile
- With AgCN → Isocyanide
- With KNO₂ → Alkyl nitrite
- With AgNO₂ → Nitro compound
- With Mg (dry ether) → Grignard reagent
- With Na (dry ether) → Alkane (Wurtz reaction)
3. Aryl Halides
- Undergo electrophilic substitution
- Nitration
- Halogenation
- Sulphonation
- Friedel–Crafts reactions
- Show ortho & para directing effect
| Factor | SN2 | SN1 |
|---|---|---|
| Kinetics | 2nd order | 1st order |
| Molecularity | Bimolecular | Unimolecular |
| Number of steps | One step | Two steps |
| Bond making & breaking | Simultaneous | Bond breaks first, then forms |
| Transition state | Pentacoordinate T.S. present | No T.S. (carbocation forms) |
| Direction of attack | Only backside attack | Front + backside attack |
| Stereochemistry | Inversion of configuration | Racemisation |
| Type of substrate | Mainly 1° | Mainly 3° |
| Polarity of solvent | Aprotic/low polarity | Polar protic |
| Intermediate | No intermediate | Carbocation intermediate |
| Reactivity order | 3° < 2° < 1° < CH₃X | 3° > 2° > 1° > CH₃X |
| Nature of nucleophile | Strong nucleophile required | Independent of nucleophile |
Concepts [12]
- Introduction of Halogen Derivatives
- Classification of Halogen Derivatives
- Nomenclature of Halogen Derivatives
- Methods of Preparation of Alkyl Halides
- Physical Properties of Halogen Derivatives
- Optical Isomerism in Halogen Derivatives
- Chemical Properties of Halogen Derivatives
- Mechanism of SN reaction
- Factors influencing SN 1 and SN 2 mechanism
- Elimination reaction: Dehydrohologenotion
- Reaction with Active Metals
- Polyhalogen Compounds
