Advertisements
Advertisements
प्रश्न
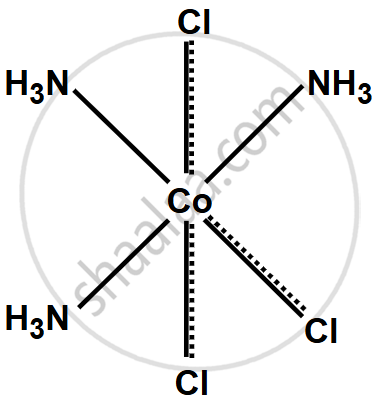
Write the structure of the following complex on the basis of Werner’s theory:
CoCl3·3NH3
रासायनिक समीकरणे/रचना
Advertisements
उत्तर
The complex CoCl3·3NH3 follows Werner’s theory, where cobalt (Co) has a +3 oxidation state (primary valency) and forms six bonds in total. Three chloride ions (Cl−) are attached through primary valency, and three ammonia molecules (NH3) are attached through secondary valency. The structure is octahedral with cobalt at the center, bonded to three chloride ions and three ammonia molecules. In solution, it ionizes as:
\[\ce{CoCl3(NH3)3 -> [Co(NH3)3]^3+ + 3Cl^-}\]
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
