Advertisements
Advertisements
प्रश्न
Write the names and structural formulae of oxoacids of chlorine.
Advertisements
उत्तर
1) Hypochlorous acid, HOCl

2) Chlorous acid, HOClO or HClO2
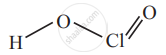
3) Chloric acid, HClO3
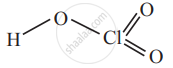
4) Perchloric acid, HClO4

APPEARS IN
संबंधित प्रश्न
Answer the following.
What is the oxidation state of ‘S’ in H2SO4?
Answer the following.
What happens when Br2 reacts with excess of F2.
Write a balanced chemical reaction of sulfuric acid with carbon.
What is the oxidation state of sulphur in the following?
Sulphurous acid
Write three uses of sulfuric acid.
Draw the structure of chloric acid.
Draw the structure of chlorous acid.
The formula of chlorous acid is ____________.
Which of the following oxyacids of sulphur contain four S = 0 bonds?
Which of the following molecular formula represents Marshall's acid?
The following structure represents

In which of the following oxoacids 'Cl' exhibit +5 oxidation state?
The molecular formula of peroxydisulfuric acid is ____________.
The number of S = O bond in peroxy monosulfuric acid is _____________.
Which of the following oxyacids of chlorine does not contain lone pair of electron on chlorine atom?
Which of the following halogen does NOT form perhalic acid?
The oxidation number of sulfur in the product is ____________.
\[\ce{I2 + SO2 + 2H2O ->}\]
Which of the following structure represents chloric acid?
Write the structure of HClO4.
Which among the following oxoacids of phosphorus shows a tendency of disproportionation?
How is chlorine obtained by oxidation of HCl. Write the reactions using two oxidizing agents.
Draw the structure of hypochlorous acid.
