Advertisements
Advertisements
प्रश्न
Write the formulae for the following compound:
Mercury(II) chloride
Advertisements
उत्तर
Mercury (II) chloride: HgCl2
APPEARS IN
संबंधित प्रश्न
EΘ values of some redox couples are given below. On the basis of these values.
EΘ values: \[\ce{Br2 /Br- = + 1.90; Ag+ / Ag(s) = + 0.80}\];
\[\ce{Cu^{2+} / Cu(s) = + 0.34; I2(s) / I- = 0.54}\]
According to the classical concept, involves one of the following?
(A) \[\ce{HOCl + H2O2 -> H3O^+ + Cl^- + O2}\]
(B) \[\ce{I2 + H2O2 + 2OH^- -> 2I^- + 2H2O + O2}\]
Choose the correct option.
Given below are two statements:
Statement I: The E° value for \[\ce{\frac{Ce^{4+}}{Ce^{3+}}}\] is +1.74 V.
Statement II: Ce is more stable in Ce4+ state than Ce3+ state.
In the light of the above statements, choose the correct answer from the options given below:
\[\ce{2MnO^-4 + {b}C2O^{2-}4 + {c}H^+ -> {x}Mn^{2+} + {y}CO2 + {z}H2O}\]
If the above equation is balanced with integer coefficients, the value of c is ______. (Round off to the Nearest Integer).
Given, that `"E"_("O"_2//"H"_2"O")^circ` = + 1.23 V;
`"E"_("S"_2"O"_8^{2-}//"SO"_4^(2-))` = 2.05 V;
`"E"_("Br"_2//"Br")^circ` = 1.09 V;
`"E"_("Au"^{2+}//"Au")^circ` = + 1.4 V
The strongest oxidizing agent is:
Ge (II) compounds are powerful reducing agents, whereas Pb (IV) compounds are strong oxidants. It can be due to ______.
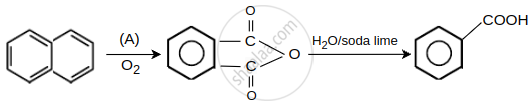
Oxidizing agent (A) used is ______.
The compound that cannot act both as oxidizing and reducing agent is ______.
Copper becomes green when exposed to moist air for a long period. This is due to ______
Which one of the following cannot function as an oxidizing agent?
Which reaction is not a redox reaction?
