Advertisements
Advertisements
प्रश्न
Write the equations for the preparation of ethyl chloride using
- Hydrogen halide
- ethene
- Thionyl chloride
Which of these methods is preferred and why?
रासायनिक समीकरणे/रचना
दीर्घउत्तर
Advertisements
उत्तर
- Hydrogen halide:
\[\ce{\underset{\text{Ethanol}}{CH3CH2 - OH + HCl} ->[anhydrous][ZnCl2] \underset{\text{Ethyl chloride}}{CH3CH2 - Cl} + H2O}\] - Ethene:
\[\ce{\underset{\text{Ethene}}{CH2 = CH2} + HCl -> \underset{\text{Ethyl chloride}}{CH3 - CH2 - Cl}}\] - Thionyl chloride:
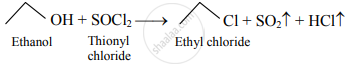
- The preparation of ethyl chloride using thionyl chloride will be the most preferred method.
1. Thionyl chloride reacts with straight-chain primary alcohols to give unrearranged alkyl chloride.
2. The byproducts (SO2 and HCl) obtained during the course of the reaction are in a gaseous state thus, there is no need to put extra effort for its separation. Hence, this method is preferred for the preparation of ethyl chloride.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
