Advertisements
Advertisements
प्रश्न
Write the chemical reaction when chlorobenzene is heated with conc. HNO3 in the presence of conc. H2SO4.
रासायनिक समीकरणे/रचना
Advertisements
उत्तर १
1. Formation of the Nitronium Ion (Electrophile): The concentrated H2SO4 acts as a catalyst to generate the active nitronium ion \[\ce{(NO^+_2)}\] from nitric acid.
\[\ce{HNO3 + 2H2SO4 -> NO^+ + 2H2SO^-_4 + H3O^+}\]
2. Nitration of Chlorobenzene: The chlorobenzene reacts with the nitronium ion to form a mixture of ortho and para-substituted products.
\[\ce{C6H5Cl ->[conc{.} H2SO4][conc{.} HNO3] \underset{o-chloronitrobenzene}{C6H4(Cl)(NO2)} + \underset{p-chloronitrobenzene}{C6H4(Cl)(NO2)} + H2O}\]
shaalaa.com
उत्तर २
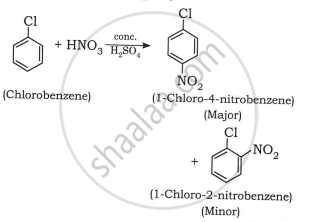
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
2025-2026 (March) Official Board Paper
