Advertisements
Advertisements
प्रश्न
Write down the formulae of magnesium hydroxide.
Advertisements
उत्तर
| Ions present | Formula |
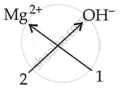 |
Mg(OH)2 |
APPEARS IN
संबंधित प्रश्न
An element Z has a valency of 3. What is the formula of oxide of Z ?
Name the elements water is made of. What are the valencies of these elements ? Work out the chemical
formula for water.
An element X has a valency of 4 whereas another element Y has a valency of 1. What will be the formula of the compound formed between X and Y ?
Give the formula and valency of:
chromate ………….…….. .
Give the empirical formula of Acitic Acid CH3COOH.
Identify the following substance:
An alkaline gas A which gives dense white fumes with hydrogen chloride.
Write the formula of the following compound:
Copper [II] oxide
State of the following formula of a compound is incorrect. Incorrectly write the correct formula.
KCr2O7
State of the following formula of a compound is incorrect. Incorrectly write the correct formula.
NaO
