Advertisements
Advertisements
प्रश्न
Write a brief note on the mechanism of enzyme catalysis.
Explain the mechanism of enzyme catalysis.
Write in brief the mechanism of enzyme catalysis.
Advertisements
उत्तर
Each enzyme produced in a particular living cell can catalyse a particular reaction in the cell.
Mechanism of enzyme catalysis: The mechanism of enzyme catalysed reaction is known as lock and key mechanism.
1. Enzymes arc highly specific in their action.
2. This specificity is due to the pressure of active sites. The shape of the active site of any given enzyme is like a cavity such that only a specific substrate can fit into it.
In the same way, a key fits into a lock. The specific binding needs to the formation of an enzyme substrate complex which accounts for the high specificity of enzyme catalysed reactions.
3. Once the proper orientation is attained the substrate molecules react to form the product in two steps.
4. Since product molecules do not have an affinity for the enzyme they leave the enzyme surface making room for the fresh substrate.
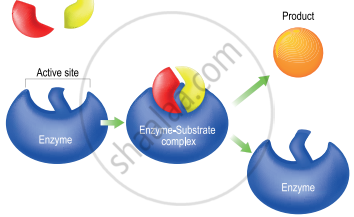
Mechanism of Enzyme Catalysis
Step 1: Formation of the enzyme-substrate complex:
\[\begin{array}{cc}
\ce{\underset{(Enzyme)}{E} + \underset{(Substrate)}{S} ⇌ \underset{(Complex)}{ES}}\\
\ce{Fast and reversible reaction}
\end{array}\]
Step 2: Dissociation of enzyme-substrate complex to form product:
\[\begin{array}{cc}
\ce{\underset{(Complex)}{ES} ⇌ \underset{(Enzyme Product Association)}{[EP]} -> \underset{(Enzyme)}{E} + \underset{(Product)}{P}}\\
\ce{Slow step reaction}
\end{array}\]
The rate of the formation of product depends upon concentration of ES.
APPEARS IN
संबंधित प्रश्न
Define the term Inhibition.
Define the term Catalysis.
In the reaction, \[\ce{H2O2_{(aq)} ->[I^-_{( aq)}] H2O_{(l)} + \frac{1}{2} O2_{(g)}}\] iodide ion acts as ____________.
171 g of sucrose on hydrolysis gives ____________ mole(s) of glucose.
(Atomic wt: C = 12, H = 1, O = 16)
Which one of the following is an example for homogeneous catalysis?
What are enzymes?
Write a note on catalytic poison.
Match List - I with List - II.
| List - I | List - II | ||
| (Processes/Reactions) | (Catalyst) | ||
| (A) | \[\ce{2SO2(g) + O2(g) -> 2SO3(g)}\] | (I) | Fe(s) |
| (B) | \[\ce{4NH3 (g) + 5O2(g) -> 4NO(g) + 6H2O (g)}\] | (II) | Pt (s) - Rh (s) |
| (C) | \[\ce{N2(g) + 3H2(g) -> 2NH3(g)}\] | (III) | V2O5 |
| (D) | \[\ce{Vegetable oil (l) + H2 -> Vegetable ghee (s)}\] | (IV) | Ni (s) |
Choose the correct answer from the options given below:
Match the catalysts to the correct processes:
| Catalysts | Process | ||
| (A) | TiCl4 | (i) | Wacker process |
| (B) | PdCl2 | (ii) | Ziegler-Natta polymerization |
| (C) | CuCl2 | (iii) | Contact process |
| (D) | V2O5 | (iv) | Deacon’s process |
In preparation of sulphuric acid from sulphur dioxide in lead chamber process. What substance is used as a catalyst?
