Advertisements
Advertisements
प्रश्न
Why are carbon and its compounds used as fuels?
Advertisements
उत्तर
The following explanations explain why fuels made of carbon and its compounds are used:
- Because of their high hydrogen and carbon content, they provide combustion with a significant amount of heat.
- Fuel-grade carbon compounds are straightforward to work with and have high calorific values and ideal ignition temperatures.
- There is control over their combustion. Carbon and its components are therefore employed as fuels.
APPEARS IN
संबंधित प्रश्न
Fill in the blank
...............is used in making black shoe polish.
Define the following:
Adsorption
How will you collect the gas ?
Write the balanced chemical equations for the preparation of carbon dioxide by:
the action of acetic acid on sodium bicarbonate.
What is a fire extinguisher ? What is the substance used in the modern type of fire extinguishers ? How is it an improvement over the soda acid-type and the foam-type fire extinguishers ?
Tick the most appropriate answer.
Each molecule of Buckminster fulierene contains 60 atoms of carbon arranged as balls consisting of 12 pentagons and
a. 20 hexagons
b. 30 hexagons
c. 40 hexagons
d. 50 hexagons
FILL IN THE BLANK
.................... is the main gaseous compound of carbon.
Carbon forms innumerable compounds by combining with other elements. True or false ?
What is the atomic number of carbon ?
How is gas carbon produced?
Name of the following compounds :

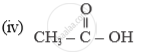
State how you would convert carbon dioxide to a metallic carbonate using a basic oxide e.g. sodium oxide. [Give a balanced equation]
Give a balanced equation for the following conversion: [In one or two steps]
Carbon dioxide to carbon.
Complete the statement by filling the blank with the correct word from the option.
Sodium oxide combines with carbon dioxide to give _____
Which of the following is highly toxic?
______ is the main constituent of coal.
Answer the following by rearranging the jumbled letters :
The reaction of carbon with oxygen gives osdiex
Answer the following by rearranging the jumbled letters :
In this molecule, carbon is attached to four hydrõgen atoms.
emathen.
Answer the following by rearranging the jumbled letters :
Carbon combines with other elements through the _______ bond.
lnaocvet.
Why is carbon monoxide harmful to health? (or) What happens when we breathe Carbon monoxide?
