Advertisements
Advertisements
प्रश्न
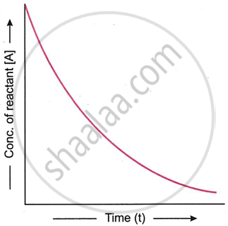
What will be the shape of the curve if the concentration of the reactant is plotted against time for a first order reaction?
आलेख
लघु उत्तर
Advertisements
उत्तर
We know that
[A] = [A]0 e−kt
According to this equation, a first-order process is exponential, and the reactant concentration drops exponentially over time. Therefore, an exponential decay curve is produced when a graph is plotted between the reactant concentrations at various points in time.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
