Advertisements
Advertisements
प्रश्न
What types of structures are possessed by ionic solids of the type AB? Explain with diagrams.
आकृती
स्पष्ट करा
Advertisements
उत्तर
- Rock Salt (NaCl) Structure: In this structure, both the A cations and B anions form a face-centred cubic (fcc) lattice. Each cation is surrounded by 6 anions, and each anion is surrounded by 6 cations, resulting in a 6 : 6 coordination. Example: NaCl, KCl, RbCl.
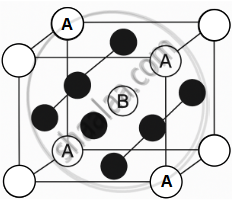
- Caesium Chloride (CsCl) Structure: In this structure, the A cations form a simple cubic lattice, while the B anions occupy the body-centred positions. Each cation is surrounded by 8 anions, and each anion is surrounded by 8 cations, giving an 8 : 8 coordination. Example: CsCl, CsBr, CsI.
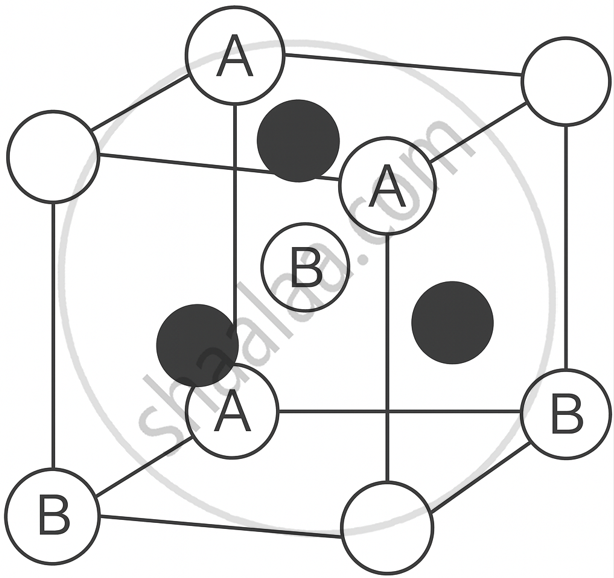 Zinc Blende (ZnS) Structure: In this structure, the B anions form a face-centred cubic (fcc) lattice, and the A cations occupy half of the tetrahedral voids. Each cation is surrounded by 4 anions, and each anion is surrounded by 4 cations, giving a 4 : 4 coordination. Example: ZnS, CuCl, AgI.
Zinc Blende (ZnS) Structure: In this structure, the B anions form a face-centred cubic (fcc) lattice, and the A cations occupy half of the tetrahedral voids. Each cation is surrounded by 4 anions, and each anion is surrounded by 4 cations, giving a 4 : 4 coordination. Example: ZnS, CuCl, AgI.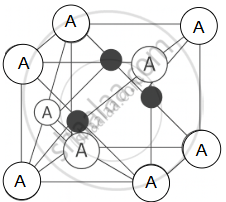
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
