Advertisements
Advertisements
प्रश्न
What observation you will record and how will you determine the specific latent heat of fusion of ice?
Advertisements
उत्तर
A clean and dry copper calorimeter with a strirrer, is weighed when empty, again weighed when filled nearly 2/3 with water. Then initial temperature is noted. Some pieces of dry ice are transferred into the calorimeter. The calorimeter is placed inside the box and is continuously stirred till the whole of ice is melted. The final temperature is carefully noted. Then the mass of calorimeter with water and ice is again weighed.
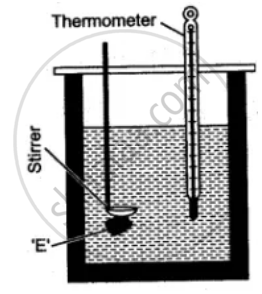
The observations recorded are:
Mass of empty calorimeter with stirrer = m1 gm.
Mass of calorimeter + water = m2 gm.
Initial temperature of water = t1 °C
Final temperature of mixture = t2 °C
Mass of calorimeter + Water + Ice (in the form of water) = m3 gm.
Temperature of ice = 0 °C
If L j/gm be the specific latent heat of fusion of ice and sc be the specific heat capacity of copper, then:
Mass of ice method = (m3 - m1) gm
Heat required by ice water at 0 °C to rise to t2 °C
= (m3 - m2) × 4.2 × (t2 - 0)
Heat lost by calorimeter = m1 × Sc × (t1 - t2)
Heat lost by water = (m2 - m1) × 4.2 × (t1 - t2)
From the heat equation,
Heat gained = Heat lost
(m3 - m2) L + (m3 - m2) × 4.2 × t2 = m1 × sc (t1 - t2) + (m2 - m1) × 4.2 × (t1 - t2)
Therefore,
L = `[(("m"_1"s"_"c")("t"_1 - "t"_2) + ("m"_2 - "m"_1) xx 4.2 xx ("t"_1 - "t"_2) - ("m"_3 - "m"_2) xx 4.2 xx "t"_2)/(("m"_3 - "m"_2))]`
