Advertisements
Advertisements
प्रश्न
What is the significance of the cell constant in the measurement of conductivity of an unknown electrolytic solution?
Advertisements
उत्तर
A conductivity cell and a Wheatstone bridge are used to test the resistance of an electrolytic solution. However, there is a significant issue with the approach. The solution electrolyses as a result of the direct current (D.C.) drawn from the battery. The solution’s concentration varies as a result of electrolysis. As a result, the solution’s resistance changes. The polarisation effect is the name given to this phenomenon. An alternating current, or A.C., is utilised to solve this issue. A suitable null point detector, such as a headphone, is used in place of a galvanometer because it cannot function with air conditioning. The conductivity cell’s electrodes are covered with finely divided platinum black to reduce the polarisation effect.
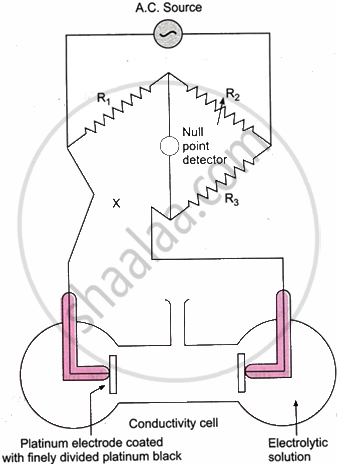
As illustrated in the figure, the electrolytic solution whose conductance needs to be measured is put inside a conductivity cell, which is then connected to the Wheatstone bridge’s fourth arm (arm AD). An A.C. source and an appropriate null point detector are used in place of the battery and galvanometer, respectively. Until the null point, where no current passes through the detector, is reached, variable resistance R2 is now changed. According to the Wheatstone bridge principle, at the null point,
`R_1/R_2 = X/R_3`
or, `X = (R_1 R_3)/R_2`
where X is the solution’s resistance. Given that conductance (C) is the opposite of resistance, we have, for the electrolytic solution in question,
Conductance (C) = `1/X`
= `R_2/(R_1 R_3)`
