Advertisements
Advertisements
प्रश्न
What is Ellingham diagram and how is it useful in metallurgical processes?
What is Ellingham diagram and how is it useful in the choice of a reducing agent in a particular metallurgical operation? Give an example.
Advertisements
उत्तर
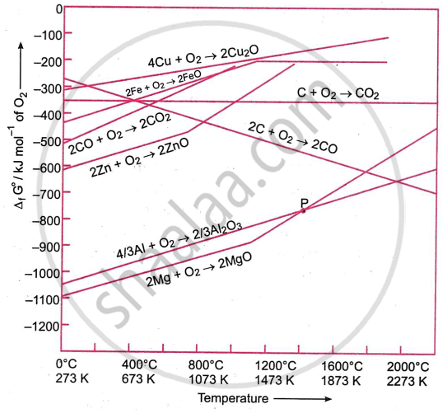
Graphical representation consisting of the plots of ΔfG° vs T for formatiion of oxides of elements is known as Ellingham Diagram.
The Ellingham diagram is extremely important in metallurgy because:
-
Prediction of Feasibility of Reduction: It helps to predict whether a metal oxide can be reduced by a given reducing agent (C, CO, H2, Al, etc.) at a particular temperature. The reaction is feasible if the line of the reducing agent lies below the line of the metal oxide (i.e., ΔG of the reducing agent is more negative).
-
Choice of Reducing Agent: For example, carbon reduces ZnO to Zn, but it cannot reduce Al2O3 because the Al2O3 line lies much lower than the C/CO line. Thus, Al is used instead of C in the thermite process.
-
Coupling of Reactions: A non-feasible reaction (ΔG > 0) can be made feasible by coupling it with another highly negative ΔG reaction (as shown in the diagram).