Advertisements
Advertisements
प्रश्न
What is a salt bridge?
Advertisements
उत्तर
A salt bridge is a U-shaped glass tube containing a saturated solution of an inert electrolyte, such as KCl or NH4NO3, Na2SO4, in a solidified agar-agar gel. A hot saturated solution of these electrolytes in 5% agar solution is filled in the U-shaped tube and allowed to cool and solidify, forming a gel.
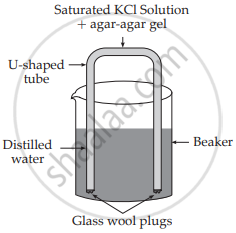
APPEARS IN
संबंधित प्रश्न
Answer the following in one or two sentences.
Write any two functions of salt bridge.
Answer the following in one or two sentences.
Formulate a cell from the following electrode reactions:
`"Au"_(("aq"))^(3+) + 3"e"^(-) -> "Au"_(("s"))`
Mg(s) → `"Mg"_(("aq"))^(2+)` + 2e-
Answer the following in one or two sentences.
What is the significance of the single vertical line and double vertical line in the formulation galvanic cell?
Calculate `"E"_"cell"^0` for galvanic cell with electrodes Co/Co3+// Mn2+/Mn.
`"E"_"Mn"^0` = - 1.18 V, `"E"_"Co"^0` = 1.82 V.
Give the chemical composition present in the salt bridge.
What are the functions of a salt bridge in a galvanic cell?
Represent the galvanic cell from following overall cell reaction:
3Ni(s) + 2Al3+ (1 M) → 3Ni2+(1 M) + 2Al(s)
Calculate standard cell potential of following galvanic cell:
Zn/Zn2+(1 M) // Pb2+(1 M)/Pb. If `"E"_"Pb"^0` = 0.126 V and `"E"_"Zn"^0` = –0.763 V
A single vertical line is used to denote __________ in the cell notation of galvanic cell.
In the reaction, \[\ce{Cu_{(s)} + 2Ag^+_{( aq)} -> Cu^{2+}_{( aq)} + 2Ag_{(s)}}\]; the reduction half-cell reaction:
Select the INCORRECT statement regarding galvanic cell notation.
The \[\ce{E^0_{cell}}\] for the following cell is:
\[\ce{Fe_{(s)} | Fe^{2+}_{( aq)} || Zn^{2+}_{( aq)} | Zn_{(s)}}\]
\[\ce{E^0_{Fe}}\] = −0.41 V,
\[\ce{E^0_{Zn}}\] = −0.76 V
Which of the following scientic notation of some figures is not correct?
Formulate the galvanic cell in which the following reaction occurs.
2Al(S) + 3Cu2+(aq) → 2Al3+(aq) + 3Cu(S)
A voltaic cell consisting of Fe2+(aq)| Fe(s) and Bi3+(aq)| Bi(s) electrodes is constructed. When the circuit is closed, mass of Fe electrode decreases and that of Bi electrode increases.
- Write cell formula .
- Which electrode is cathode and which electrode is anode?
- Write electrode reactions and overall cell reaction.
Write two uses of salt bridge.
Which of the following is selected as the cathode for a galvanic cell set up with a nickel anode?
