Advertisements
Advertisements
प्रश्न
What happens when Nitrobenzene undergoes electrolytic reduction in a strongly acidic medium?
Advertisements
उत्तर
Electrolytic reduction of nitrobenzene in weakly acidic medium gives aniline but in strongly acidic medium, it gives p-amino phenol obviously through the acid catalysed rearrangement of the initially formed phenyl hydroxylamine.

APPEARS IN
संबंधित प्रश्न
Which one of the following will not undergo Hofmann bromamide reaction?
\[\ce{CH3CH2Br ->[aq NaOH][\Delta] A ->[KMnO4/H^+][\Delta] B ->[NH3][\Delta] C ->[Br2/NaOH] D}\] ‘D’ is:
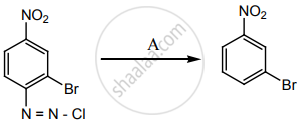
‘A’ is:
What happens when 2-Nitropropane boiled with HCl?
What happens when oxidation of tert-butylamine with KMnO4?
How will you convert nitrobenzene into o and p-nitrophenol?
How will you convert nitrobenzene into hydrozobenzene?
How will you convert nitrobenzene into aniline?
Account for the following.
Aniline does not undergo Friedel–Crafts reaction
Account for the following.
Aniline does not undergo Friedel – Crafts reaction.
