Advertisements
Advertisements
प्रश्न
What do you observe when caustic soda solution is added to the following solution: first a little and then in excess : FeCl3
Advertisements
उत्तर
Addition of caustic soda to FeCl3 solution :
\[\ce{FeCl3 + 3NaOH ->\underset{\text{Reddish Brown ppt.}}{Fe(OH)3 v + 3NaCI}}\]
APPEARS IN
संबंधित प्रश्न
Choose the most appropriate answer from the following list of oxides which fit the description.
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
A basic oxide.
Choose the most appropriate answer from the following list of oxides which fit the description
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
An amphoteric oxide
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
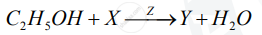
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Give a chemical test to distinguish between the given pairs of chemicals:
Lead nitrate solution and Zinc nitrate solution
Name the metal hydroxides which are :
(a) Insoluble (b) Soluble in
(i) Caustic soda solution (ii) Ammonium hydroxide solution.
Name: A weak alkali.
Name a compound containing zinc in the anion.
Choose the correct answer:
The metal oxide which can react with acid as well as alkali is:
Write balanced equations for a metal that evolves into a gas which burns with a pop sound when boiled with alkali solutions.
Write balanced equation for a metal that evolves a gas which burns with a pop sound when boiled with alkali solution.
