Advertisements
Advertisements
प्रश्न
The energy levels of an atom of a certain element are shown in the given figure. Which one of the transitions A, B, C, D or E will result in the emission of photons of electromagnetic radiation of wavelength 618.75 nm? Support your answer with mathematical calculations.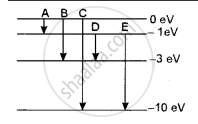
बेरीज
Advertisements
उत्तर
The wavelength emitted λ = 618.75 nm
∴ Corresponding energy E = h `c/λ`
= `( 6.6 xx 10^-34 xx 3 xx 10^8 )/( 618.75 xx 10^-9 xx 1.6 xx 10^-19) e^V`
= `( 6.6 xx 3 )/( 618.75 xx 1.6 ) xx 100`
= 2eV
This corresponds to the transition D.
In which energy emitted is ΔE = - 1 - ( - 3 ) = 2eV
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
