Advertisements
Advertisements
प्रश्न
The adsorption of gases on solids can be described by Freundlich’s equation, `x/m = kP^(1//n)`.
- How are the values of k and n determined experimentally?
- Explain, why should the value of n be equal to or greater than one?
स्पष्ट करा
Advertisements
उत्तर
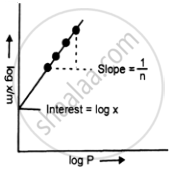
Fig. Linear graph between `bb(log (x/m))` and log P.
Taking logarithms on both sides, we get
\[\log\left(\frac{x}{m}\right) = \log k + \frac{1}{n} \log P\]
Thus, if we plot a graph between `log (x/m)` and log P, a straight line will be obtained. The slope of the line is equal to `1/n` and the intercept is equal to log k. Thus the values of k and n are determined. n can take a value any where between 1 and a large number depending upon the pressure. At low pressure n = 1.
∴ `x/m = kP`
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
