Advertisements
Advertisements
प्रश्न
Study the table given below. Use only the letters given in the table to answer the questions. Do not identify the elements.
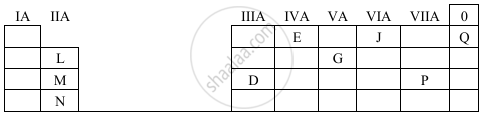
- State the valency of element ‘G’.
- Which element can exhibit catenation?
- Write the formula of the compound formed between ‘M’ and ‘P’.
दीर्घउत्तर
Advertisements
उत्तर
- G possesses five electrons in its outermost shell since it belongs to group V A. The valency of G is −3.
- As the element in group IV A exhibits catenation, group E may also exhibit it.
- As M is in group II A, its valency is +2, and because P is in group VII A, its valency is −1. The compound between M and P has the formula MP2.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
2024-2025 (March) Official Board
