Advertisements
Advertisements
प्रश्न
Show by a graphic diagram how at a constant pressure a rise in temperature will influence adsorption of a gas on a solid when (i) no compound formation occurs and (ii) chemisorption takes place.
Advertisements
उत्तर
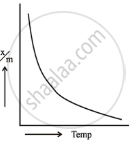
Adsorption is an exothermic reaction. Le-chatlier’s Principle predicts that adsorption increases with decreasing temperature. Physical adsorption occurs at low temperatures due to the strong van der Waals forces. However, as the temperature rises, chemisorption increases and then decreases. The initial increase demonstrates that chemisorption, like chemical reactions, requires activation energy.
 |
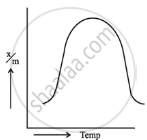 |
| Physical adsorption | Chemical adsorption |
Plotting the amount of gas adsorbed `(x/m)` vs temperature at constant equilibrium pressure reveals that physical adsorption decreases with increasing temperature. Chemisorption exhibits an initial spike followed by a consistent reduction. Such curves are referred to as adsorption isobars.
