Advertisements
Advertisements
प्रश्न
Rohit took two different salt solutions in test tubes C and D as shown in the figure below. He added dilute HCl to each of the two test tubes. The products formed in the test tubes C and D are silver chloride and lead chloride respectively.
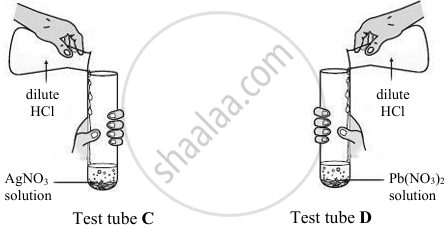
State:
- one common observation made by Rohit in both the reactions.
- the observations made by him on addition of excess of ammonium hydroxide to the products formed in:
- test tube C
- test tube D
दीर्घउत्तर
Advertisements
उत्तर
- One common observation is that a white precipitate is formed in both test tubes.
- When the addition of excess ammonium hydroxide to test tube C, the white precipitate dissolves as silver chloride dissolves in the presence of excess ammonium hydroxide, resulting in a clear solution.
- Since lead chloride only dissolves in hot water, test tube D remains unchanged when an excessive amount of ammonium hydroxide is added.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
2024-2025 (March) Official Board
