Advertisements
Advertisements
प्रश्न
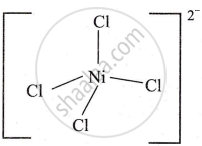
On the basis of valence bond theory, explain the nature of bonding in the [Ni(Cl)4]2− complex ion.
स्पष्ट करा
Advertisements
उत्तर
- Oxidation state of nickel is +2.
- Valence shell electronic configuration of Ni2+
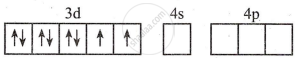
- The number of Cl− ligands is 4. Therefore, the number of vacant metal ion orbitals required for bonding with ligands must be four.
- Four orbitals on metal available for hybridisation are one 4s and three 4p. The complex is tetrahedral.
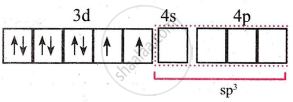
The four metal ion orbitals for bonding with Cl− ligands are derived from the sp3 hybridisation. - Four vacant sp3 hybrid orbitals of Ni2+ overlap with four orbitals of Cl− ions.
- Configuration after complex formation would be
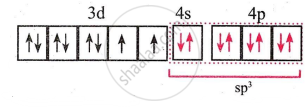
- The complex has two unpaired electrons and hence is paramagnetic.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
