Advertisements
Advertisements
प्रश्न
Name two coloured metal ions.
Advertisements
उत्तर १
Fe2+, Mn2+
उत्तर २
Cupric (Blue) [Cu2+], Ferric (Yellow) [Fe3+]
APPEARS IN
संबंधित प्रश्न
Choose the most appropriate answer from the following list of oxides which fit the description
[SO2, SiO2, Al2O3, MgO, CO, Na2O]
An amphoteric oxide
A compound X (having the vinegar-like smell) when treated with ethanol in the presence of the acid Z, gives a compound Y which has a fruity smell.
The reaction is:
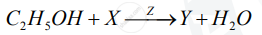
1) Identify Y and Z.
2) Write the structural formula of X.
3) Name the above reaction.
Name two colourless metal ions.
What do you understand by amphoteric oxide? Give the balanced equations for the reaction with three different amphoteric oxides with a caustic alkali. Write your observation if any.
Give two examples of amphoteric hydroxides.
Fill in the blank.
The hydroxide which is soluble in excess of NaOH is _________ [Zn(OH)2 / Fe(OH)3 / Fe(OH)2].
Fill in the blank.
The substance/s which react with hot conc. NaOH solution and undergoes a neutralization reaction ______ [Al2O3 / Al / Al(OH)3]
Fill in the blank.
To distinguish soluble salts of zinc and lead ______ [NaOH / NH4OH]can be used.
When ammonium hydroxide is added to solution B, a pale blue precipitate is formed. This pale blue precipitate dissolves in excess ammonium hydroxide to give an inky blue solution. Name the cation present in solution B. What is the probable colour of solution B?
What do you see when sodium hydroxide solution is added to zinc sulphate solution, till it is in excess?
