Advertisements
Advertisements
प्रश्न
(i)
Write the mathematical expression relating to the variation of the rate constant of a reaction with temperature.
(ii)
How can you graphically find the activation energy of the reaction from the above expression?
(iii)
The slope of the line in the graph of log k (k = rate constant) versus `1/"T"` is -5841. Calculate the activation energy of the reaction.
थोडक्यात उत्तर
Advertisements
उत्तर
(i)
`"k" = "Ae"^(- ("E"_"a")/("RT"))`
k = rate constant
A = collision factor
Ea = activation energy
(ii)
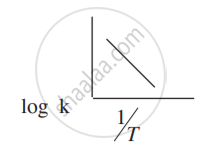
Slope = -`"E"_"a"/(2.303"R")`
(iii)
∴ Ea = – 2.303 × slope × R
= – 2.303 × – 5841 × 8.314
= 111838.4 J mol-1
= 111.8 KJ mol-1
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
