Advertisements
Advertisements
प्रश्न
How would you determine the activation energy of a reaction with the help of Arrhenius equation?
How can activation energy be determined for a reaction with the help of Arrhenius equation?
Advertisements
उत्तर
Graphical method:
\[\ce{log_10 k = - \frac{E_a}{2.303 RT} + log_10 A}\]
This is an equation of the type y = mx + c and represents a straight line. Therefore, if the values of k are plotted against \[\ce{\frac{1}{T}}\], a straight line should be obtained, and the slope of this line should be equal to \[\ce{- \frac{E_a}{2.303 R}}\].
Therefore, in order to determine the energy of ·activation of a reaction, its rate constants at different temperatures are measured. Now, the values of log10 k against \[\ce{\frac{1}{T}}\] are plotted.
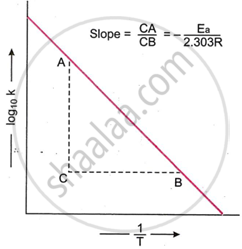
The curve obtained is a straight line, as shown in the figure. The slope of the line is measured. The slope of the line is related to the activation energy of the reaction as follows.
\[\ce{Slope = \frac{CA}{CB} = - \frac{E_a}{2.303 R}}\]
Rate constant method:
If k1 and k2 are the rate constants measured at temperatures T1 and T2, respectively, then according to \[\ce{log_10 k = - \frac{E_a}{2.303 RT} + log_10 A}\], we have
\[\ce{log_10 k_1 = - \frac{E_a}{2.303 RT_1} + log_10 A}\] ...(i)
\[\ce{log_10 k_2 = - \frac{E_a}{2.303 RT_2} + log_10 A}\] ...(ii)
Subtracting eq. (i) from eq. (ii), we get
\[\ce{log_10 k_2 - log_10 k_1 = \frac{E_a}{2.303 R} ({\frac{1}{T_1}} - {\frac{1}{T_2}})}\]
or, \[\ce{log_10 \frac{k_2}{k_1} = \frac{E_a}{2.303 R} (\frac{1}{T_1} - \frac{1}{T_2})}\] ...(iii)
Equation (iii) can therefore be used to determine the energy of activation (Ea) of a reaction given the values of its rate constants, k1 and k2, measured at two distinct temperatures, T1 and T2, respectively.
