Advertisements
Advertisements
प्रश्न
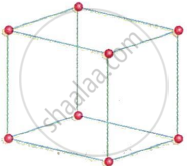
How many atoms of an element are there in one unit cell of simple cubic?
How many atoms are present per unit cell in a primitive unit cell?
Calculate the number of particles per unit cell in a simple cubic system.
संख्यात्मक
Advertisements
उत्तर
Simple cubic systems contain primitive unit cells. In a primitive unit cell (also called a simple cubic unit cell), only the corners of a cube contain the basic particles, such as atoms. There are eight corners on a cube. Since eight unit cells share each corner atom, each corner atom contributes 1/8 to the unit cell. Thus, the quantity of atoms in a single basic cubic unit cell = `8 xx 1/8 = 1`. A simple cubic unit cell contains 1 atom of the element.

shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
