Advertisements
Advertisements
प्रश्न
How is the instantaneous rate of a reaction determined at a particular instant? Explain graphically.
Advertisements
उत्तर
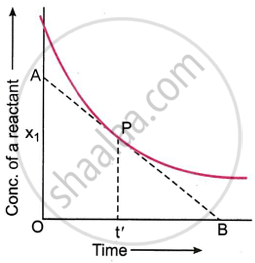
The rate of reaction at a specific moment in time is referred to as the instantaneous rate. It can be ascertained by calculating the slope of the concentration vs. time curve at a certain point that corresponds to the rate determination time.
The figure shows a typical curve that is produced when a reactant's measured concentration is plotted against time. Suppose the instantaneous rate of the reaction at time t' is to be determined. Let this time be represented by point P on the curve. A tangent AB is drawn at point P so that it crosses the time axis at point B and the concentration axis at point A. This allows one to determine the slope of the curve at that point.
The change in concentration Δx is given by the distance OA along the concentration axis, while the change in time Δt is given by the distance OB along the time axis. The slope of the curve at time t' is provided by the ratio OA/OB. Thus, the instantaneous rate of reaction at time t' = slope of the curve at point p = \[\ce{\frac{OA}{OB}}\].
