Advertisements
Advertisements
प्रश्न
How is copper extracted from its ores?
Advertisements
उत्तर
Copper is extracted mainly from copper pyrites. The extraction is carried out in the following steps.
Crushing and concentration: Jaw crushers are used to crush the large ore particles, while ball mills are used to finely powder the material. The method of froth flotation is used to concentrate the powdered ore.

As shown in the figure, the finely crushed ore slurry (ore + water) is placed in a sizable tank for this procedure. The following materials were additionally added to the ore slurry.
- Frothing agents: Pine or eucalyptus oil is used as a foaming agent. The froth remains steady and rises to the tank’s surface.
- Collectors: Ethyl xanthate or potassium ethyl xanthate is employed as the collector. This enhances the binding force between ore particles and froth. It attaches to polar groups in mineral grains. The ore particles become water-repellent and pass into the froth.
- Activators or depressants: These chemicals activate or repress flotation in ore containing several minerals.
Roasting: Concentrated ore is roasted in a reverberatory furnace with a free air supply. The alterations listed below occur during roasting.
- Impurities in the ore, including sulphur, arsenic, and antimony, undergo oxidation and are removed as volatile oxides.
\[\ce{S + O2 -> SO2 \uparrow}\]
\[\ce{4As + 3O2 -> 2As2O3 \uparrow}\]
\[\ce{4Sb + 3O2 -> 2Sb2O3 \uparrow}\] - A mixture of ferrous and cuprous sulphides is produced from the ore.
\[\ce{2CuFeS2 + O2 -> Cu2S + 2FeS + SO2}\] - The resulting sulphides undergo partial oxidation to their corresponding lower oxides.
\[\ce{2CuS + 3O2 -> 2Cu2O + 2SO2}\]
\[\ce{2FeS + 3O2 -> 2FeO + 2SO2}\]
Smelting: Coke, sand, and roasted ore are combined and then put into a tiny blast furnace. The furnace is about 5-7 mins height and about 1-1.5 m in diameter and is made of steel lined inside with fire bricks.
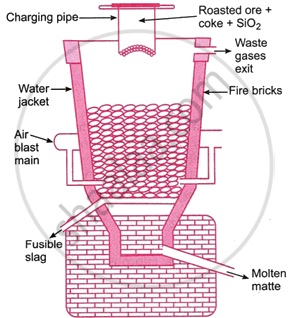
The shallow soil at the furnace’s base has distinct tapping holes for the slag and matte. A charging line and a waste gas outlet are located on top of the furnace. A burst of hot air is sent via the tuyeres at the base, just over the hearth, to heat the furnace.
Bessemerisation: A Bessemer converter receives the matte that was produced in the blast furnace. It is a steel pear-shaped furnace with a simple lime or magnesium oxide lining within.
It may be tilted in any direction for charging and discharging and is supported by trunnions. It has tuyeres, or pipes, installed in the sides that are high enough above the bottom. The metal that is created falls below the tuyeres, preventing it from being oxidised by the air blast.
