Advertisements
Advertisements
प्रश्न
How does the molar conductivity of a weak electrolytic solution vary with concentration?
Why is it not possible to determine `Lambda_m^infty` for a weak electrolyte by the usual methods? Explain graphically.
Describe the characteristics of variation of conductance with dilution for a weak electrolyte. How would you explain it qualitatively?
How does the molar conductivity of a weak electrolyte vary with concentration? Explain graphically. Why is it not possible to determine `Lambda_m^infty` for a weak electrolyte graphically?
Advertisements
उत्तर
Weak electrolytes, such as CH3COOH, NH4OH, and HCN, dissociate less in solution than strong electrolytes. As a result, a weak electrolyte’s solution contains many fewer ions than a strong electrolyte of the same concentration. As a result, a weak electrolyte solution has significantly lower molar conductivity than a strong electrolyte.
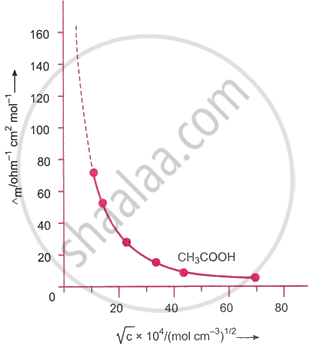
The molar conductivity of a weak electrolyte grows gradually at lower concentrations before quickly increasing as the concentration of the solution is decreased (i.e., dilution is raised). As a result, a weak electrolyte’s molar conductivity does not change linearly with concentration. Figure displays the graph that plots the values of acetic acid’s molar conductivity (Λm) against the square root of its concentration`(sqrt c)`. It is evident from this graph that the curve is not a straight line, indicating that for a weak electrolyte, the change in molar conductivity with concentration is nonlinear.
