Advertisements
Advertisements
प्रश्न
How does a fuel cell operate?
Advertisements
उत्तर
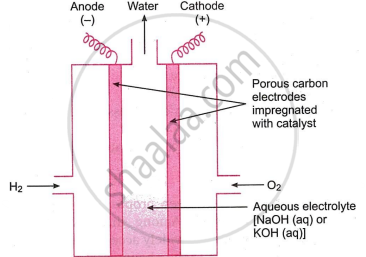
The cells that convert the chemical energy of a fuel directly into electrical energy are called fuel cells.
Bacon created the hydrogen-oxygen fuel cell in 1959. As a result, another name for it is Bacon cell. In the Apollo space program of the United States, it served as the main source of electrical energy and has the capacity to do so.
Fig. shows a basic H2-O2 fuel cell. Two porous carbon electrodes loaded with an appropriate catalyst, such as Pt, Ag, CoO, etc., make up the cell. An electrolyte, which is a concentrated solution of KOH or NaOH, fills the gap between the two electrodes. As illustrated in the fig., H and O gases bubble in the electrolyte via porous carbon electrodes. Reactions occur while the cell is functioning.