Advertisements
Advertisements
प्रश्न
How can a galvanic cell be constructed?
Advertisements
उत्तर
In a galvanic cell, a redox reaction is always the chemical mechanism that produces electrical energy. Therefore, an oxidation electrode and an appropriate reduction electrode can be used to construct a galvanic cell. Either a salt bridge (Fig. b) or direct contact via a porous diaphragm (Fig. a) can bring the solution in the two electrodes into communication with the other. The two electrodes are connected to a device in the outer circuit that can use the electrical energy produced.
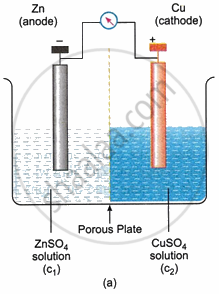
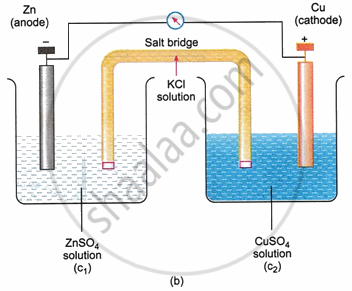
The oxidation process at the oxidation electrode allows the electrons to be liberated. These electrons build up on the electrode and give it a negative potential if they are not removed. A positive voltage arises at the reduction electrode as a result of the reduction process taking place there. Due to a potential difference between the two electrodes, electrons begin moving from the oxidation electrode to the reduction electrode in the outer circuit when they connect together (in both the inner and outer circuits). Therefore, an electric current is produced.
The electrode at which the oxidation process occurs is called the Anode while the one at which the reduction process takes place is called the Cathode. It is to be noted that the anode is of negative polarity, while the cathode is of positive polarity in a galvanic cell.
