Advertisements
Advertisements
प्रश्न
Give a balanced equation for the following conversion: [In one or two steps]
A metallic oxide to calcium carbonate.
Advertisements
उत्तर
A metallic oxide to calcium carbonate
Step I: \[\ce{\underset{\text{Cal.oxide}}{CaO} + H2O -> Ca[OH]2}\]
Step II: \[\ce{Ca[OH]2 + CO2 ->\underset{\text{[Cal. carbonate]}}{CaCO3} + H2O}\]
APPEARS IN
संबंधित प्रश्न
Which of the following molecule is called buckministerfullerene?
C90 C60 C70 C120
Fill in the blank
................. is present in both living and non-living things.
Choose the correct alternative.
Carbon forms innumerable compounds because
Name the products formed when:
bone is heated in the absence of air.
What are hydrocarbons ? Name the two different forms of hydrocarbons.
Name of the following compounds :

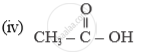
Differentiate between the two branches of chemistry – organic chemistry & inorganic chemistry with suitable examples.
State how carbon occurs in the free state and in the combined state.
State in brief the transformation of vegetable matter to different types of coal varying in carbon content.
About 18% of the weight of the human body is carbon.
