Advertisements
Advertisements
प्रश्न
Explian the following:
Dil. HCl acid is stronger than highly concentrated acetic acid.
Advertisements
उत्तर
The strength of an acid is the measure of the concentration of hydronium ions it produces in its aqueous solution. Dil. HCl produces a higher concentration of hydronium ions than concentrated acetic acid. Thus, dil. HCl is a stronger acid than highly concentrated acetic acid.
APPEARS IN
संबंधित प्रश्न
Nitric acid turns red litmus blue.
One of the following is not an organic acid. This is:
Give a balanced equation for reaction of nitrogen dioxide with water.
Give one example in the following case:
A hydrogen containing compound which is not an acid.
Tartaric acid is a ______ acid.
What are the uses of acids?
Fill in the cross word given in figure with the help of the clues provided.
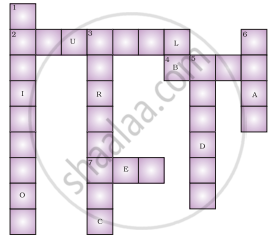
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Write the important uses of hydrochloric acid and sulphuric acids.
Explain the following:
\[\ce{H3PO3}\] is not a tribasic acid.
You are supplied with five solutions: A, B, C, D and E with pH values as follows: A = 1.8, B = 7, C = 8.5, D = 13, and E = 5
Classify these solutions as neutral, slightly or strongly acidic and slightly or strongly alkaline.
Which solution would be most likely to liberate hydrogen with powdered zinc metal. Give a word equation for each reaction.
