Advertisements
Advertisements
प्रश्न
Explain with examples the term reduction potential.
स्पष्ट करा
Advertisements
उत्तर
When the metal ion gains n electrons from the electrode, it gets reduced into metal atom M, as shown below.
\[\ce{M{^{n+}_{(aq)}} + ne- -> M_{(s)}}\]
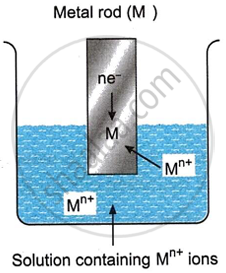
As a result, the metal ion is deposited as an atom on the metal rod’s surface. Until the accumulated charge prevents further activity, the process keeps on. A positive voltage forms on the electrode as a result of this reduction process. This form of electrode assembly is known as a reduction electrode, and the potential that is thus created is known as a reduction potential.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
