Advertisements
Advertisements
प्रश्न
Explain the term ‘linkage isomerism’ with respect to coordination compounds and illustrate with two examples.
Advertisements
उत्तर
This isomerism arises when the ligands have two different atoms available for coordinating with the metal ion, i.e., ligands are ambident. One donor atom of the ligand coordinates with the central metal atom in one isomer, whereas the second donor atom coordinates with the metal atom in the other isomer. Example of linkage isomerism:
1. [Co(NO2)(NH3)5]2+ and [Co(ONO)(H2O)5]2+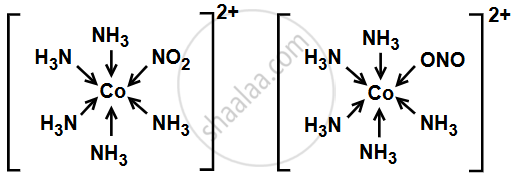
\[\ce{NO^-_2}\] (Nitrite ion) is an ambidentate ligand, meaning it can bind to the metal ion through two different atoms: nitrogen (N) or oxygen (O).
- [Co(NO2)(NH3)5]2+ : The nitrogen (N) atom of the nitrite ion is coordinating with the central cobalt ion.
- [Co(ONO)(H2O)5]2+: The oxygen (O) atom of the nitrite ion is coordinating with the cobalt ion.
These are linkage isomers, as the same nitrite ion is coordinating in different ways (through either nitrogen or oxygen).
2. [Co(SCN)(NH3)5]2+ and [Co(NCS)(H2O)5]2+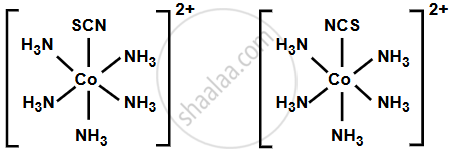
SCN− (thiocyanate ion) is also an ambidentate ligand, and it can bind through either sulfur (S) or nitrogen (N).
- [Co(SCN)(NH3)5]2+: The sulfur (S) atom of the thiocyanate ion is coordinating with the central cobalt ion.
- [Co(NCS)(H2O)5]2+: The nitrogen (N) atom of the thiocyanate ion is coordinating with the cobalt ion.
Again, these are linkage isomers, as the SCN− ligand is coordinated differently through either sulfur or nitrogen.
