Advertisements
Advertisements
प्रश्न
Explain the structure and magnetic behaviour of hexaamminenickel (II) ion on the basis of this theory.
Advertisements
उत्तर
The complex has an octahedral geometry where a central Nickel (II) ion (Ni2+) is surrounded by six ammonia (NH3) molecules acting as ligands. These ligands are positioned at the corners of an octahedron around the central metal ion. The central metal ion is nickel in the +2 oxidation state, with an electronic configuration of [Ar]3d8. Ammonia (NH3) molecules act as ligands, donating electron pairs to the Ni2+ ion to form coordinate covalent bonds.
Nickel atomic number (Z): 28
Ni: [Ar] 3d8 4s2
Ni2+: [Ar] 3d8 (after losing two 4s electrons)
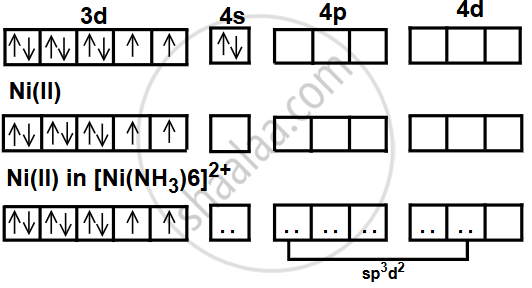
Since there are two unpaired electrons, the species is paramagnetic and since it is sp3d2 hybridization therefore shape is octahedral.
Magnetic behaviour of the hexaamminenickel (II) ion on the basis of VBT theory:
- Ammonia (NH3) is considered a weak field ligand for nickel (II).
- In the presence of weak field ligands, the crystal field splitting energy (Δo) is small compared to the pairing energy (P.E.), which is the energy required to pair up electrons in the same orbital.
- As a result, electrons will preferentially occupy the eg orbitals before pairing up in the t2g orbitals to minimize repulsion.
- The 3d8 electronic configuration of Ni2+ in an octahedral, weak field environment leads to:
- Six electrons filling the t2g orbitals (t2g).
- Two electrons occupying the eg orbitals (eg).
- This filling pattern results in two unpaired electrons in the eg orbitals.
- The presence of unpaired electrons makes the hexaamminenickel (II) ion paramagnetic, meaning it is weakly attracted to an external magnetic field.
- The magnetic moment (μ) can be estimated using the spin-only formula: μ = `sqrt(n(n+2)) BM`, where 'n' is the number of unpaired electrons.
- For [Ni(NH3)6]2+, with two unpaired electrons (n = 2), the calculated spin-only magnetic moment is approx. 2.83 BM. This is consistent with experimental values found in the range of 2.8 - 3.3 BM for octahedral Ni(II) complexes.
- Therefore, the hexaamminenickel (II) ion is a paramagnetic, high-spin, outer orbital complex.
