Advertisements
Advertisements
प्रश्न
Explain the second law of thermodynamics in terms of entropy.
Advertisements
उत्तर
The quantity `"Q"/"T"` is called entropy. It is a very important thermodynamic property of a system.
It is also a state variable. `"Q"_"H"/"T"_"H"` is the entropy received by the Carnot engine from a hot reservoir and `"Q"_"L"/"T"_"L"` is entropy given out by the Carnot engine to the cold reservoir. For reversible engines (Carnot Engine) both entropies should be the same so that the change in entropy of the Carnot engine in one cycle is zero. But for all practical engines like diesel and petrol engines which are not reversible engines, they satisfy the relation `"Q"_"L"/"T"_"L" > "Q"_"H"/"T"_"H"`.
In fact, we can reformulate the second law of thermodynamics as follows
“For all the processes that occur in nature (irreversible process), the entropy always increases. For reversible process entropy will not change”. Entropy determines the direction in which natural processes should occur.
Entropy increases when heat flows from hot objects to cold objects. If heat were to flow from a cold to a hot object, entropy will decrease leading to violation of second law thermodynamics. Entropy is also called ‘measure of disorder’. All-natural process occurs such that the disorder should always increase.
Consider a bottle with gas inside. When the gas molecules are inside the bottle it has less disorder. Once it spreads into the entire room it leads to more disorder. In other words, when the gas is inside the bottle the entropy is less and once the gas spreads into the entire room, the entropy increases. From the second law of thermodynamics, entropy always increases. If the air molecules go back into the bottle, the entropy should decrease, which is not allowed by the second law of thermodynamics. The same explanation applies to a drop of ink diffusing into water. Once the drop of ink spreads, its entropy is increased. The diffused ink can never become a drop again. So the natural processes occur in such a way that entropy should increase for all irreversible processes.
APPEARS IN
संबंधित प्रश्न
A gas contained in a cylinder surrounded by a thick layer of insulating material is quickly compressed has there been a transfer of heat?
Answer in brief.
What sets the limits on the efficiency of a heat engine?
Draw a p-V diagram and explain the concept of positive and negative work. Give one example each.
The figure shows the V-T diagram for one cycle of a hypothetical heat engine which uses the ideal gas. Draw the p-V diagram diagram of the system.
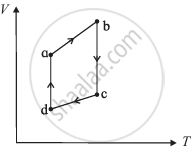
Define heat engine.
What are the processes involves in a Carnot engine?
A Carnot engine whose efficiency is 45% takes heat from a source maintained at a temperature of 327°C. To have an engine of efficiency of 60% what must be the intake temperature for the same exhaust (sink) temperature?
Which statement is incorrect?
Draw a neat labelled P - V diagram for a typical heat engine.
The thermal efficiency of a heat engine is 25%. If in one cycle the heat absorbed from the hot reservoir is 50000 J, what is the heat rejected to the cold reservoir in one cycle?
