Advertisements
Advertisements
प्रश्न
Explain the following term:
Energy barrier
स्पष्ट करा
Advertisements
उत्तर
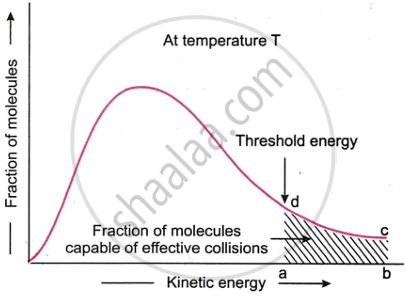
Until the reacting molecules have a specific minimum amount of energy known as the threshold energy, their collisions cannot be successful. This indicates that only when two reacting molecules have energy greater than or equal to the threshold energy will their collisions be effective and result in the production of products. Fig. shows the energy distribution between colliding molecules at a specific temperature. The percentage of molecules with energy above or equal to the threshold energy that can collide effectively is shown by the turned area abed.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
