Advertisements
Advertisements
प्रश्न
Explain in detail Newton’s law of cooling.
Advertisements
उत्तर
Newton’s law of cooling: Newton’s law of cooling states that the rate of loss of heat of a body is directly proportional to the difference in the temperature between that body and its surroundings.
`"dQ"/"dt" ∝ ("T" - "T"_"s")` ............(1)
The negative sign indicates that the quantity of heat lost by liquid goes on decreasing with time. Where,
T = Temperature of the object
Ts = Temperature of the surrounding
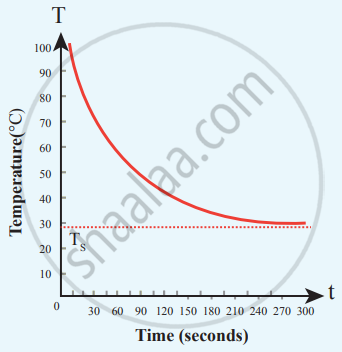
Cooling of hot water with time
From the graph in the figure, it is clear that the rate of cooling is high initially and decreases with falling temperature.
Let us consider an object of mass m and specific heat capacity s at temperature T. Let Ts be the temperature of the surroundings. If the temperature falls by a small amount dT in time dt, then the amount of heat lost is,
dQ = msdT .........(2)
Dividing both sides of equation (2) by dt
`"dQ"/"dt" = "msd"/"dt"` .........(3)
From Newton’s law of cooling `"dQ"/"dt" ∝ ("T" - "T"_"s")`
`"dQ"/"dt" = - "a"("T" - "T"_"s")` .............(4)
Where a is some positive constant. From equation (3) and (4)
− a (T − Ts) = `"ms" "dT"/"dt"`
`"dT"/("T" - "T"_"s") = -"a"/"ms" "dt"` .......(5)
Integrating equation (5) on both sides,
`int "dT"/("T" - "T"_"s") = - int "a"/"ms" "dt"`
ln (T − Ts) = `"a"/"ms" "t" + "b"_1`
Where b1, is the constant of integration. Taking exponential both sides, we get
T = `"T"_"s" + "b"_2"e"^(-"a"/"ms""t")` ........(6)
here b2 = `"e"^("b"_1)` = constant
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Water falls from a hight of 200 m. What is the difference in temperature between the water at the top and bottom of a waterfall given that the specific heat of water is 4200 J kg-1 °C-1?
Define specific heat capacity.
Write the unit of latent heat capacity.
Define thermal conductivity.
Discuss various modes of heat transfer.
A man starts bicycling in the morning at a temperature around 25°C, he checked the pressure of tire which is equal to be 500 kPa. Afternoon he found that the absolute pressure in the tyre is increased to 520 kPa. By assuming the expansion of tyre is negligible, what is the temperature of tyre at afternoon?
Another name for thermal energy is ______
What does the temperature of a body tell us?
Which of the following is the fourth fundamental quantity introduced while studying heat?
What is the difference between temperature and heat energy?
