Advertisements
Advertisements
प्रश्न
Explain briefly seven types of unit cells.
Advertisements
उत्तर
Seven types of unit cells:

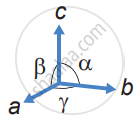
Cubic
a = b = c
α = β = γ = 90°
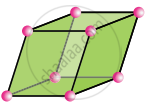
Rhombohedral
a = b = c
α = β = γ ≠ 90°

Hexagonal
a = b ≠ c
α = β = 90°, γ = 120°
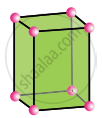
Tetragonal
a = b ≠ c
α = β = γ = 90°
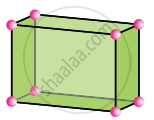
Orthorhombic
a ≠ b ≠ c
α = β = γ = 90°
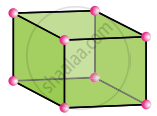
Monoclinic
a ≠ b ≠ c
α = γ = 90°, β ≠ 90°
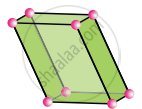
Triclinic
a ≠ b ≠ c
α ≠ β ≠ γ ≠ 90°
- Cubic – NaCl
- Rhombohedral – Cinnabar Cubic
- Hexagonal – ZnO
- Tetragonal – TiO2
- Orthorhombic – BaSO4
- Monoclinic – PbCrO4
- Triclinic – H3BO3
They differ in the arrangements of their crystallographic axes and angles.
Corresponding to the above seven, Bravis defined 14 possible crystal systems as shown in the figure.
APPEARS IN
संबंधित प्रश्न
The number of unit cells in 8 gm of an element X (atomic mass 40) which crystallizes in bcc pattern is (NA is the Avogadro number)
CsCl has bcc arrangement, its unit cell edge length is 400 pm, it's inter atomic distance is
The vacant space in bcc lattice unit cell is ____________.
The fraction of total volume occupied by the atoms in a simple cubic is
If ‘a’ stands for the edge length of the cubic system; sc, bcc, and fcc. Then the ratio of radii of spheres in these systems will be respectively.
If ‘a’ is the length of the side of the cube, the distance between the body-centered atom and one corner atom in the cube will be ______.
What is meant by the term “coordination number”?
What is the coordination number of atoms in a bcc structure?
An element has bcc structure with a cell edge of 288 pm. The density of the element is 7.2 g cm−3. How many atoms are present in 208 g of the element?
An atom crystallizes in fcc crystal lattice and has a density of 10 g cm−3 with unit cell edge length of 100 pm. calculate the number of atoms present in 1 g of crystal.
