Advertisements
Advertisements
प्रश्न
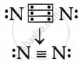
Draw the electron dot structure for the following:
A molecular nitrogen
[At. No. : N = 7, H = 1]
Advertisements
उत्तर
Nitrogen atoms share three electrons, making a triple bond.
APPEARS IN
संबंधित प्रश्न
Covalent bond is formed by ______
Polar solvents contain bonds between atoms with ______.
______ & ______ atoms have similar electronegativities.
Spot the error/correct the wrong statement:
Covalent compounds are soluble in polar solvents.
Covalent compounds have a low melting point.
Draw the electron dot structure of Methane molecule.
[Atomic number: N = 7, C = 6, H = 1]
Draw the electron dot structure of Nitrogen molecule.
[Atomic number: N = 7, C = 6, H = 1]
Carbon tetrachloride is a ______ covalent molecule.
Draw the structural formula of carbon tetrachloride and state the type of bond present in it.
Select the right answer.
Carbon tetrachloride
