Advertisements
Advertisements
प्रश्न
Describe the construction and working of a Daniell cell.
Advertisements
उत्तर
Construction: A Daniell cell consists of two half-cells. A zinc rod dipped in zinc sulphate (ZnSO4) solution forms the anode. Copper rod dipped in copper sulphate (CuSO4) solution forms the cathode. The two solutions are connected by a salt bridge (e.g., KCl or KNO3 in agar gel), which maintains electrical neutrality by allowing the flow of ions. The zinc and copper rods are connected by a wire through which electrons flow, and a voltmeter may be placed in the circuit to measure cell potential.
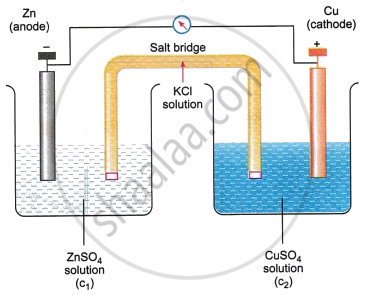
Working: At the anode (zinc electrode), oxidation occurs
\[\ce{Zn_{(s)} -> Zn{^{2+}_{(aq)}} + 2e-}\]
Zinc loses electrons and goes into solution as Zn2+ ions.
At the cathode (copper electrode), reduction occurs
\[\ce{Cu{^{2+}_{(aq)}} + 2e- -> Cu_{(s)}}\]
Cu2+ ions gain electrons and deposit as solid copper on the copper electrode.
Electrons flow from zinc to copper through the external circuit, generating an electric current.
The salt bridge allows movement of ions to balance charges; anions move toward the anode and cations toward the cathode.

Net cell reaction:
\[\ce{Zn_{(s)} + Cu{^{2+}_{(aq)}} -> Zn{^{2+}_{(aq)}} + Cu_{(s)}}\]
