Advertisements
Advertisements
प्रश्न
Define the term calorie. How is it related to joule (the S.I. unit of heat)?
Advertisements
उत्तर
One calorie of heat is the heat energy required to raise the temperature of 1 g of water from 14.5°C to 15.5°C.
1 calorie = 4.186 J
APPEARS IN
संबंधित प्रश्न
Name the three modes of transfer of heat.
Name the mode of transfer of heat in the following :
(a) solid,
(b) liquid,
(c) gas
(d) vacuum
While deciding the unit for heat, which temperature interval is chosen? Why?
What will absorb heat?
Water
Give a scientific reason.
In winter, why does an iron pillar feel colder than a wooden pole?
The unit of heat and temperature are the same.
The process of converting a liquid into a solid is called ________.
When an electric current is passing through the conductor ______ energy is produced.
A circular metal loop is heated at point O as shown in the following figure.
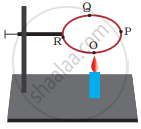
- In which direction would heat flow in the loop?
- In which order are the pins at points P, Q and R fixed with the help of wax fall if points O, P, Q and R are equidistant from each other?
