Advertisements
Advertisements
प्रश्न
Consider two containers A and B containing identical gases at the same pressure, volume and temperature. The gas in container A is compressed to half of its original volume isothermally while the gas in container B is compressed to half of its original value adiabatically. The ratio of final pressure of gas in B to that of gas in A is ______.
पर्याय
`2^(γ - 1)`
`(1/2)^(γ - 1)`
`(1/(1 - γ))^2`
`(1/(γ - 1))^2`
Advertisements
उत्तर
Consider two containers A and B containing identical gases at the same pressure, volume and temperature. The gas in container A is compressed to half of its original volume isothermally while the gas in container B is compressed to half of its original value adiabatically. The ratio of final pressure of gas in B to that of gas in A is `underline(2^(γ - 1))`.
Explanation:
According to the P-V diagram is shown for container A (which is going through an isothermal process) and for container B (which is going through an adiabatic process).
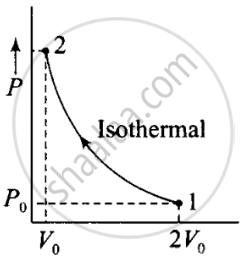
V →container A

V →container A
Both processes involve compression of the gas.
(i) Isothermal compression (gas A) (during 1→ 2)
P1V1 = P2V2
⇒ P0(2V0)γ = P2(V0)γ
⇒ P0(2V0) = P2(V0)
(ii) Adiabatic compression, (gas B) (during 1→ 2)
P1V1γ = P2V2γ
⇒ P0(2V0)γ = P2(V0)γ
⇒ P2 = `((2V_0)/V_0) P_0 - (2)^γ P_0`
Hence `((P_2)_B)/((P_2)_A)` = ratio of final pressure = `((2)^γ P_0)/(2P_0) = 2^(γ - 1)` where, γ is ratio of specific heat capacities for the gas.
