Advertisements
Advertisements
प्रश्न
Consider the section of the periodic table given below :
| Group numbers |
IA | IIA | IIIA | IVA | VA | VIA | VIIA | O |
| 1 | 2 | 13 | 14 | 15 | 16 | 17 | 18 | |
| Li | D | O | J | Ne | ||||
| A | Mg | E | Si | H | K | |||
| B | C | F | G | L |
With reference to the table, answer the following :
(i) Which is the most electro negative ?
(ii) How many valence electrons are present in G ?
(iii) Write the formula of the compound between B and H ?
(iv) In the compound between F and J, what type of bond will be formed ?
(v) Draw the electron dot structure for the compound formed between C and K.
एका वाक्यात उत्तर
Advertisements
उत्तर
(i) K is the most electro negative.
(ii) 5 valence electrons are present in G.
(iii) B2H
(iv) Ionic
(v)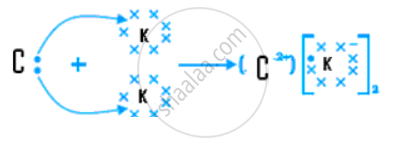
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
