Advertisements
Advertisements
प्रश्न
Classify the following solutions as acids, bases or salts.
Ammonium hydroxide, barium chloride, sodium chloride, sodium hydroxide, H2SO4 and HNO3
Advertisements
उत्तर
Acids: H2SO4 and HNO3
Bases: Ammonium hydroxide and sodium hydroxide.
Salts: Barium chloride and sodium chloride.
APPEARS IN
संबंधित प्रश्न
What happens to the H+ ions, of the solution, when an acid is neutralised?
Which chemical is injected into the skin of a person during the nettle leaf hair sting?
How can the effect of these stings be neutralised?
Name an acid used as an eyewash.
Name the particles present in :
Weak electrolyte
Give the name and formula of two : Non-volatile acids
Explain, all alkalis are bases but all bases are not alkalis.
Most of the acids are not soluble in water.
Fill in the cross word given in figure with the help of the clues provided.
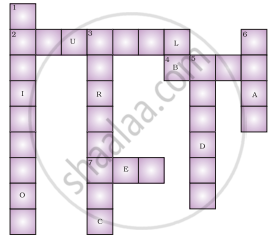
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Which one of the following acids undergoes complete ionisation?
Bring the soil sample of your area, find out if it is acidic, basic or neutral. Discuss with farmers if they treat the soil in any manner.
