Advertisements
Advertisements
प्रश्न
Chlorine trifluoride molecule undergoes sp3d hybridization, excited state electronic configuration of chlorine atom can be written as:
पर्याय
1s2 2s2 2p6 3s2 3p4 3d1
1s2 2s2 2p6 3s2 3p3 3d2
1s2 2s2 2p6 3s2 3p5
1s2 2s2 2p6 3s1 3p5 3d1
MCQ
Advertisements
उत्तर
1s2 2s2 2p6 3s2 3p4 3d1
Explanation:
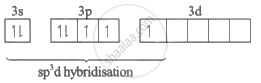
CIF3 = sp3d hybridization
Cl = 3s2 3p5
Excited State of Cl: 1s2 2s2 2p6 3s2 3p4 3d1
shaalaa.com
Hybridization
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
