Advertisements
Advertisements
प्रश्न
Can the adsorption of a solute from a solution be explained on the basis of the Freundlich adsorption isotherm? Explain.
Advertisements
उत्तर
Yes, the adsorption of a solute from a solution can be explained on the basis of the Freundlich adsorption isotherm.
According to this empirical relationship,
`x/m = KC^(1//n)`
Where:
`x/m` = Amount of solute adsorbed per unit mass of adsorbent
C = Equilibrium concentration of the solute in solution
K and n = Constants depending on the nature of the adsorbent and solute and the temperature.
Taking the logarithm on both sides:
`log(x/m) = log K + 1/n log C`
This equation is of the form y = mx + c, representing a straight line.
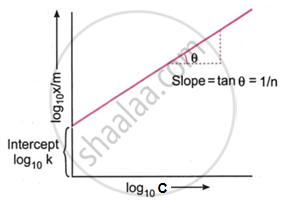
A plot of `log(x/m)` logC gives a straight line.
Slope = `1/n`
Intercept = log K
This confirms the validity of the Freundlich isotherm for solute adsorption from solution.
