Advertisements
Advertisements
प्रश्न
Answer the following.
Explain in detail free radical mechanism involved during preparation of addition polymer.
Advertisements
उत्तर
The free radical mechanism is most common in addition to polymerization. It is also called a chain reaction which involves three distinct steps. These are as follows:
i) Step 1: Chain initiation:
a. The chain reaction is initiated by a free radical which is formed from an initiator (catalyst) such as benzoyl peroxide, acetyl peroxide, tert-butyl peroxide, etc.
b. For example, acetyl peroxide generates methyl radical as shown below:

c. The free radical (say `"R"^•`) so formed attaches itself to the olefin (vinyl monomer) and produces a new radical, made up of two parts, namely, the attached radical and the monomer unit.

ii. Step 2: Chain propagation:
a. The new radical formed in the initiation step reacts with another molecule of vinyl monomer, forming another still bigger sized radical, which in turn reacts with another monomer molecule.
b. The repetition of this sequence takes place very rapidly. It is called chain propagation.
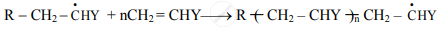
c. This step is very rapid and leads to high molecular mass radical.
iii. Step 3: Chain termination:
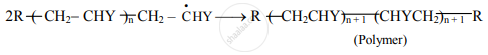
a. At some stage, termination of the growing chain takes place. It may occur by several processes.
b. One mode of termination is by combination of two growing chain radicals.
APPEARS IN
संबंधित प्रश्न
Answer the following in one sentence.
Define the term: Synthetic fibres
Answer the following
How is polythene manufactured? Give their properties and uses.
Attempt the following:
Classify the following polymer as straight-chain, branched-chain and cross-linked polymers.
\[\begin{array}{cc}
\ce{-(CH2 - CH2 - CH - CH2 - CH2)_\text{n}}\\
|\phantom{..}\\\ce{\phantom{..}CH2}\\
|\phantom{..}\\\ce{\phantom{..}CH2}\end{array}\]
Semisynthetic polymer among the following is ______________
Homopolymer among the following is ______.
Which of the following is a biodegradable polymer?
Chemical combination of Ziegler-Natta catalyst is ______________
Functional group present in terylene polymer is ______.
Define elastomer.
Explain thermoplastic and thermosetting polymers.
Explain the classification of polymers on the basis of structures.
Which of the following properties is of the thermosetting polymers?
Identify the monomers used in the preparation of glyptal.
The total number of electrons around the carbon atom of methyl tree radical are ______.
Which among the following is not a semi-synthetic polymer?
Which of the following pair represents natural polymers?
Which of the following is INCORRECT regarding terylene?
Which of the following compounds contain -CO-NH- linkage?
Which among the following polymer is used to make crockeries?
In the following reaction, the reason for why meta-nitro product also formed is:

Identity semisynthetic polymer from following.
Write names of two natural polymers.
Which from following is a thermoplastic polymer?
Identify thermoplastic polymer from following.
